Water Tension: What's Holding it All Together
Now, it is time to discuss one of the most unique features about water: water tension!
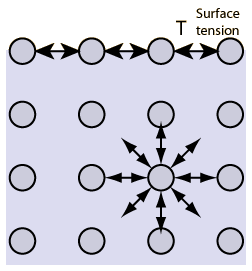
Water has a unique property known as Surface Tension. What does this mean?
o The molecules of a liquid experience cohesive attractive forces from all directions
o On the surface, molecules are drawn inward towards the water’s center
This means that the surface of water sticks holds together, similar to bouncing on a trampoline. The surface of water contracts like a stretched, elastic membrane. If an object is light enough, such as many species of bugs, it is possible to walk on top of water. Water density contributes to this as well.
Water has a unique property known as Surface Tension. What does this mean?
o The molecules of a liquid experience cohesive attractive forces from all directions
o On the surface, molecules are drawn inward towards the water’s center
This means that the surface of water sticks holds together, similar to bouncing on a trampoline. The surface of water contracts like a stretched, elastic membrane. If an object is light enough, such as many species of bugs, it is possible to walk on top of water. Water density contributes to this as well.
Ok...so what? What does this really do?
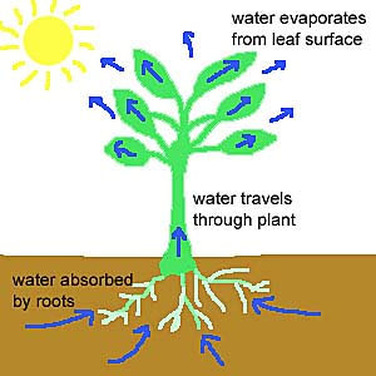
This actually does quite a lot. According to Cohesion-tension theory, “water sticks together and can move up a plant from the roots to the leaves”. The tension, this attractive force between water molecules, allows for upward movement of water molecules up plant cells. water molecules pull each other along up from a plant's roots to its leaves, where it can get absorbed by the atmosphere.
This actually does quite a lot. According to Cohesion-tension theory, “water sticks together and can move up a plant from the roots to the leaves”. The tension, this attractive force between water molecules, allows for upward movement of water molecules up plant cells. water molecules pull each other along up from a plant's roots to its leaves, where it can get absorbed by the atmosphere.